
2025-11-03 Posted by TideChem view:781
Since the initial discovery and successful use of insulin for treating type 1 diabetes in 1922, therapeutic peptides have undergone over a century of development. Owing to their high affinity, high selectivity, low toxicity, and broad range of indications, peptide drugs have garnered extensive attention and achieved remarkable progress.
Despite their advantages, peptide drugs face considerable challenges: inherent instability leading to protease degradation, combined with low molecular weight causing rapid renal clearance. This results in poor oral bioavailability, short half-life, and frequent dosing requirements that severely impact patient compliance and drug development.
To address this, half-life extension technologies have emerged. These strategies—including non-natural amino acid substitution, fusion with long-acting fragments, fatty acid acylation, PEGylation, and sustained-release formulations—can significantly prolong the circulation time of peptide drugs in vivo and optimize their overall pharmacokinetic profile. Currently, these technologies are widely employed in the development and improvement of various therapeutic peptides.
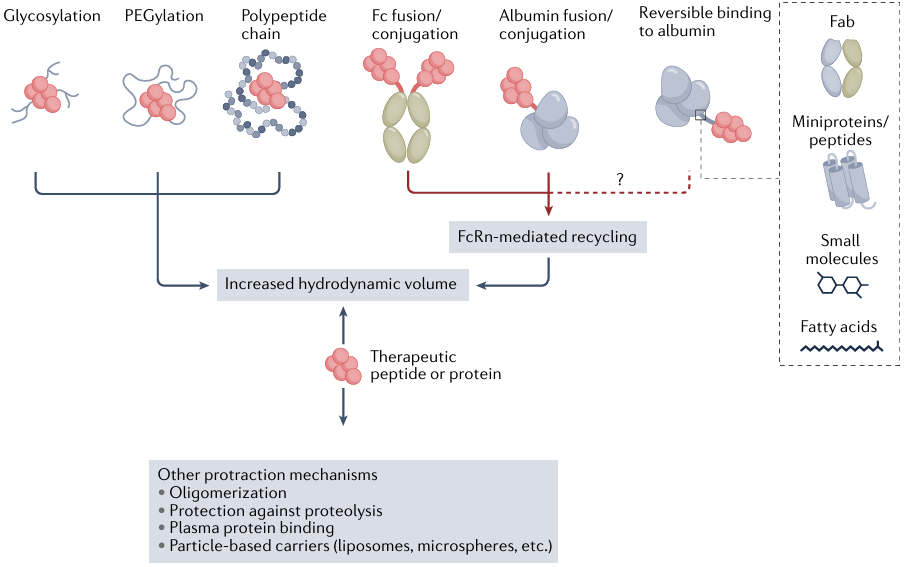
Figure 1. Technologies for half-life extension of therapeutic peptides.
The human gastrointestinal tract contains abundant proteolytic enzymes, which hydrolyze the peptide bonds of therapeutic peptides, leading to their rapid clearance or conversion into active or inactive metabolites. These metabolic processes occur extensively and rapidly, thereby resulting in an extremely short in vivo half-life of peptide drugs.
The incorporation of non-natural amino acids can effectively enhance the chemical stability and enzymatic resistance of peptide drugs, protecting their peptide backbone from degradation and hydrolysis, thereby increasing the drug's half-life and bioavailability, and extending its duration of action in vivo.
For instance, native somatostatin is rapidly degraded by enzymes in human plasma, exhibiting a plasma half-life of merely 1–2 minutes. In contrast, its analog, octreotide, incorporates two D-amino acid residues (D-Trp and D-Phe) at key positions. Since the peptide bonds formed by D-amino acids exhibit greater resistance to proteolytic enzymes, the stability of octreotide is significantly enhanced, resulting in an extended plasma half-life of 1.5 hours while retaining pharmacological activity comparable to that of the native hormone.
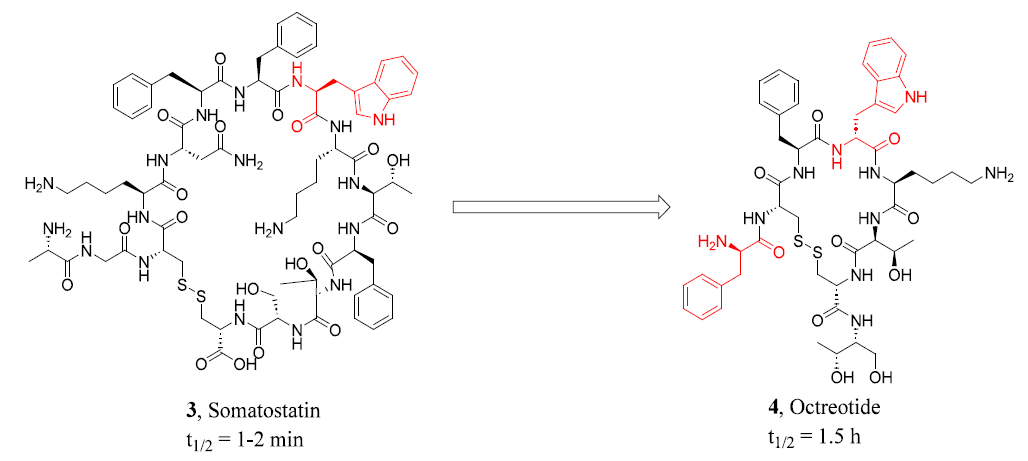
Figure 2. Structures of somatostatin and octreotide.
IgG and human serum albumin (HSA) are the two most abundant proteins in human blood. Their long half-life of approximately 20 days makes them ideal carriers for improving the pharmacokinetic profiles of therapeutic drugs. This prolonged circulation time is primarily attributed to two factors: first, their large molecular size effectively prevents rapid glomerular filtration; second, both can be efficiently recycled back into the bloodstream via an intracellular recycling mechanism mediated by the neonatal Fc receptor (FcRn), thereby significantly extending their retention time in the circulation.
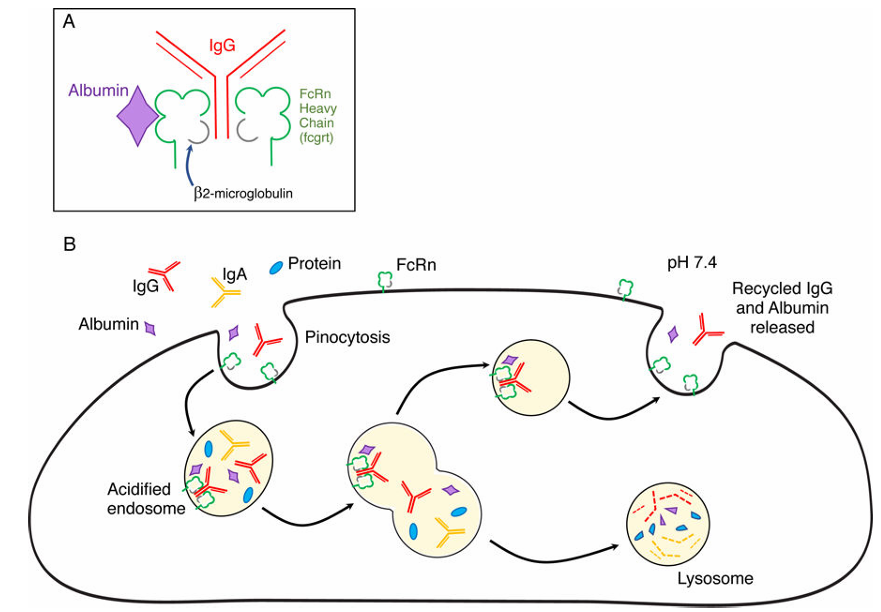
Figure 3. The structure and function of FcRn.
Human GLP-1 is a peptide composed of 37 amino acid residues. It is susceptible to rapid enzymatic degradation and renal clearance in vivo, resulting in a plasma half-life of merely 1–2 minutes, which severely restricts its clinical applicability.
Albiglutide was developed by GlaxoSmithKline and received FDA approval in 2014 for treating type 2 diabetes. It is a recombinant human serum albumin (HSA) molecule, comprising two copies of GLP-1(7-36) fused to its N-terminus. The sequence incorporates an A8G substitution that confers enhanced resistance to degradation by dipeptidyl peptidase-4 (DPP-4), while fusion to HSA extends its half-life in humans to 6-8 days, thereby enabling a once-weekly subcutaneous dosing regimen.
Dulaglutide was developed by Eli Lilly and Company as a long-acting GLP-1 receptor agonist and also received FDA approval in 2014. Its design incorporates two critical structural modifications: first, amino acid substitutions within the GLP-1 moiety to reduce its susceptibility to cleavage by dipeptidyl peptidase-4 (DPP-4); second, a covalent linkage of its C-terminus to the N-terminus of a human IgG4-Fc segment via a 16-amino-acid peptide linker. This design effectively reduces renal clearance, extends the plasma half-life to approximately 4 days, enables once-weekly dosing, and significantly improves patient adherence.
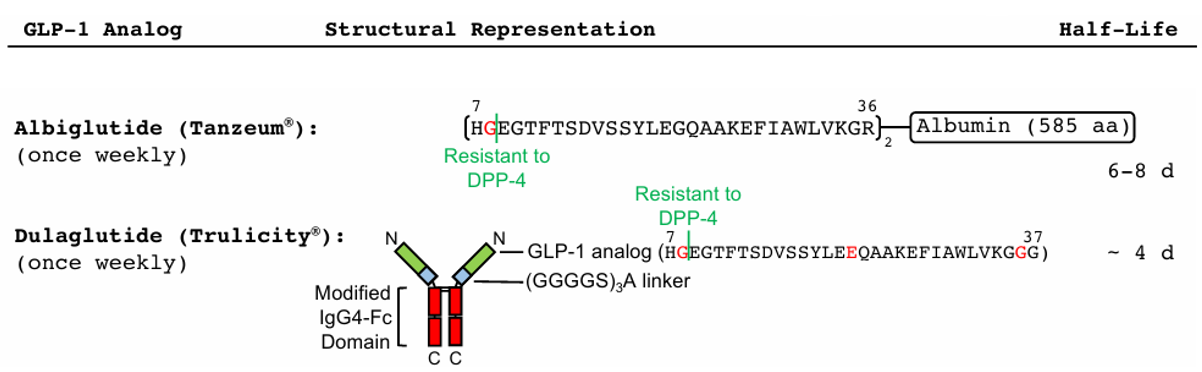
Figure 4. Structures of Albiglutide and Dulaglutide.
Fatty acid acylation technology significantly enhances the pharmacokinetic properties of peptides by covalently conjugating hydrophobic fatty acid chains to the peptide backbone, and has been successfully applied in several marketed peptide drugs. The long-acting mechanism primarily involves three aspects: first, the fatty acid side chain can efficiently and reversibly bind to human serum albumin in vivo, forming an "albumin-peptide complex" reservoir that enables sustained drug release; second, the increased molecular size of the complex effectively avoids rapid renal filtration, thereby delaying clearance; additionally, albumin binding shields certain proteolytic cleavage sites on the peptide chain, enhancing its stability against protease degradation. These mechanisms work synergistically to significantly prolong the plasma half-life of peptide drugs.
A series of therapeutics utilizing fatty acid derivatization technology have been translated into clinically approved therapies, including Somapacitan for growth hormone deficiency, Zilucoplan for myasthenia gravis, as well as Semaglutide and Tirzepatide for obesity and type 2 diabetes. Furthermore, a number of new candidates based on this technology are currently under active development, indicating its potential for broader therapeutic application in the future and underscoring the remarkable versatility and promising prospects of this technological platform.
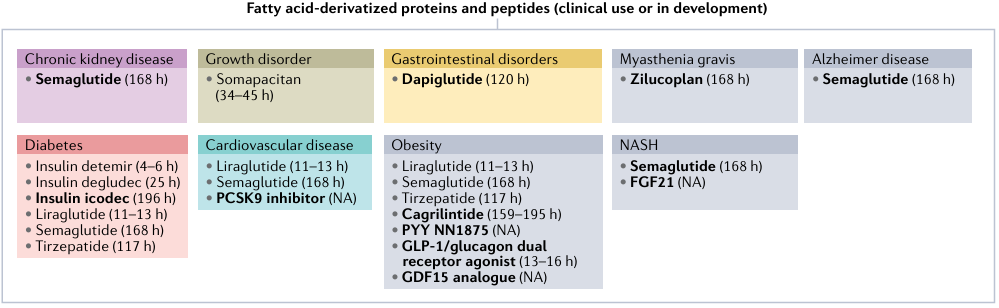
Figure 5. Fatty acid-derivatized proteins and peptides (clinical use or in development).
Polyethylene glycol (PEG) is a water-soluble polymer with excellent biocompatibility. When used to modify peptide drugs, PEGylation significantly increases the drug's molecular weight, thereby reducing renal filtration and clearance. Simultaneously, the PEG chains shield proteolytic cleavage sites on the peptide surface, enhancing its resistance to enzymatic degradation. Moreover, the "stealth" property of PEG helps reduce the immunogenicity of the drug. Through these dual mechanisms of physical shielding and biological regulation, PEGylation effectively extends the plasma half-life of peptide drugs and has become one of the most mature and successful strategies for peptide half-life extension.
Pegcetacoplan targets complement C3 protein and is indicated for treating paroxysmal nocturnal hemoglobinuria (PNH) and other complement-mediated disorders, having received FDA accelerated approval in 2021. The peptide structure features an intramolecular disulfide bond between two cysteine side chains, while the terminal Thr is conjugated to a PEG chain via an AEEA spacer and Lys linker, extending plasma half-life to enable biweekly administration.
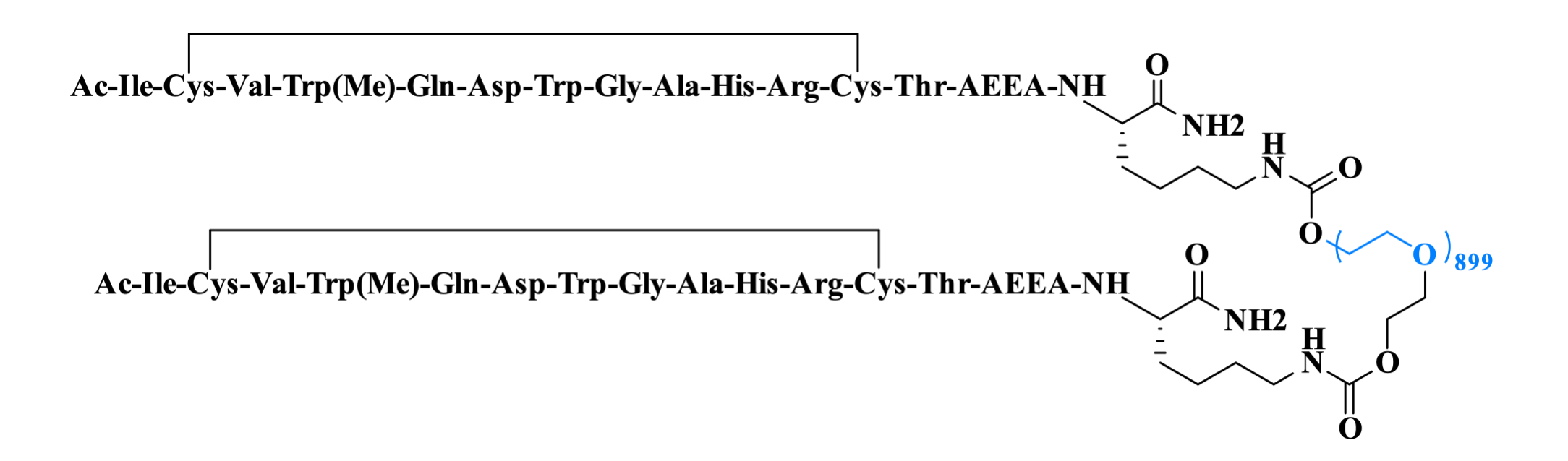
Figure 6. The structure of Pegcetacoplan.
Microsphere formulations are typically administered via subcutaneous or intramuscular injection, achieving sustained drug release through controlled degradation of the carrier material. This delivery platform offers high biocompatibility and targeting efficiency, making it an ideal vehicle for peptide therapeutics.
A prominent example is AstraZeneca's exenatide extended-release microsphere product (marketed as Bydureon®), which employs PLGA as its matrix. This formulation converts the original twice-daily subcutaneous injection regimen to a convenient once-weekly administration, substantially enhancing patient treatment adherence.
Tide Chem specializes in the field of peptide drug prolongation technologies, offering a comprehensive portfolio of hundreds of structurally diverse non-natural amino acids, fatty acid side chains, and PEG linkers. Our capabilities fully support projects from milligram-scale exploration to kilogram-scale production, effectively meeting clients' customized requirements throughout the entire peptide drug development cycle.
Binder, Uli, and Arne Skerra. “Strategies for extending the half-life of biotherapeutics: successes and complications.” Expert opinion on biological therapy vol. 25,1 (2025): 93-118. doi:10.1080/14712598.2024.2436094.
Zaman, Rahela et al. “Current strategies in extending half-lives of therapeutic proteins.” Journal of controlled release : official journal of the Controlled Release Society vol. 301 (2019): 176-189. doi:10.1016/j.jconrel.2019.02.016.
Sharma, Krishna K et al. “Unnatural Amino Acids: Strategies, Designs, and Applications in Medicinal Chemistry and Drug Discovery.” Journal of medicinal chemistry vol. 67,22 (2024): 19932-19965. doi:10.1021/acs.jmedchem.4c00110.
Baldwin, William M 3rd et al. “The neonatal Fc receptor: Key to homeostasic control of IgG and IgG-related biopharmaceuticals.” American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons vol. 19,7 (2019): 1881-1887. doi:10.1111/ajt.15366.
Cheang, Jia Ying, and Peter M Moyle. “Glucagon-Like Peptide-1 (GLP-1)-Based Therapeutics: Current Status and Future Opportunities beyond Type 2 Diabetes.” ChemMedChem vol. 13,7 (2018): 662-671. doi:10.1002/cmdc.201700781.
Kurtzhals, Peter et al. “Derivatization with fatty acids in peptide and protein drug discovery.” Nature reviews. Drug discovery vol. 22,1 (2023): 59-80. doi:10.1038/s41573-022-00529-w.