
2025-12-26 Posted by TideChem view:337
Peptide and protein drugs, as bioactive molecules composed of amino acids, are characterized by high specificity, low toxicity, and favorable biocompatibility, making them a critical area in pharmaceutical research. With continuous advancements in biotechnology and peptide synthesis techniques, an increasing number of peptide and protein drugs have been successfully applied in clinical practice. Currently, these drugs have demonstrated significant therapeutic efficacy in treating a wide range of diseases, including cancer, diabetes, obesity, growth disorders, and myasthenia gravis.
However, many natural peptide and protein drugs are prone to enzymatic hydrolysis or rapid renal clearance, leading to low oral bioavailability and short in vivo half-lives. To address these limitations, chemical modifications and recombinant technologies are commonly employed to prolong the duration of drug action by increasing molecular size or leveraging the neonatal Fc receptor (FcRn) recycling mechanism. Among these approaches, fatty acid acylation technology stands as one of the most successful and widely applied strategies for extending the half-life of peptide and protein drugs.
Fatty acid-derived peptide or protein drugs can undergo oligomerization at the injection site, forming multimers that increase molecular size, slow subcutaneous absorption rate, and significantly extend plasma half-life. For example:
Human insulin is a glucose-lowering peptide hormone secreted by pancreatic pancreatic β-cells, consisting of 51 amino acids formed by the A and B chains linked via disulfide bonds. In its natural form, insulin has a short half-life—approximately 5–15 min after intravenous injection and 2–5 h after subcutaneous administration. Insulin degludec is acylated at the ε‑NH2 of Lys29 via a γ‑glutamic acid-linked C16 diacid (HO‑C16‑Glu(OSU)‑OH, CAS: 943586‑12‑7). This modification enables it to form stable multi-hexamer structures at the injection site. These multimers gradually release monomers into the bloodstream, extending its intravenous half-life to about 5 h and subcutaneous half-life up to 25 h. This significantly reduces dosing frequency and provides patients with a more convenient treatment regimen.
Liraglutide, as a GLP-1 analog, is acylated at the ε‑NH2 group of Lys26 via γ‑glutamic acid linked to a C16 monoacid (Pal‑Glu(OSU)‑OH, CAS: 294855‑91‑7). Its subcutaneous injection half-life is markedly extended from 1–2 min for native GLP‑1 to 11–13 h, enabling once‑daily administration. This derivative forms tightly bound heptamers in the formulation buffer. After injection, the strong interactions between monomers allow slow release and subsequent binding to albumin, creating a long‑acting depot that prolongs its circulation time in vivo.
Unlike the mechanisms of insulin degludec and liraglutide, which rely on multimer formation, the GLP‑1 agonist semaglutide utilizes a C18 fatty diacid (Ste‑Glu‑AEEA‑AEEA‑OSU, CAS: 1169630‑40‑3) to achieve potent binding to albumin. This increases the molecular weight and shields enzymatic cleavage sites, while simultaneously reducing renal clearance through the FcRn‑mediated recycling pathway, enabling once‑weekly subcutaneous administration.1
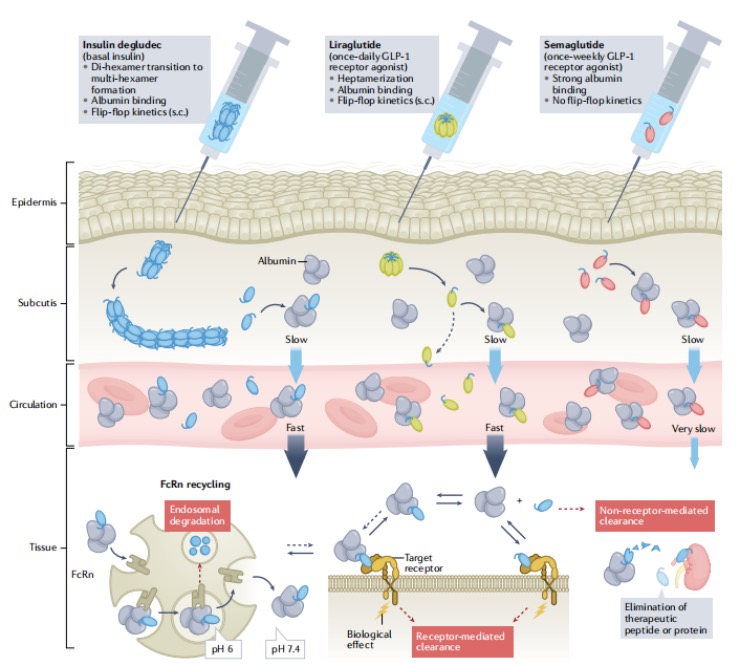
Fig1.The Mechanism of Action of Fatty Acid Derivatization
Fatty acid derivatization technology is a core strategy for extending the half-life of peptide and protein drugs. This technique involves chemically attaching fatty acid side chains of varying structures to drug molecules, thereby significantly improving their pharmacokinetic properties.
Compared to fatty acids containing only one carboxyl group at the alkyl chain terminus (monoacids), fatty acids with an additional carboxyl group at the ω-terminus (diacids) generally exhibit stronger binding affinity to albumin, and the albumin-binding capacity of both types increases with the length of the alkyl chain. Research has shown that the C18 diacid (Fmoc-L-Lys[Oct-(OtBu)-Glu-(OtBu)-AEEA-AEEA]-OH, CAS: 1662688-20-1) and the C20 diacid side chain (Fmoc-Lys[AEEA-AEEA-γ-Glu(OtBu)-C20-OtBu]-OH, CAS: 2915356-76-0) 2 exhibit the highest affinity for albumin. Additionally, fatty diacids are more hydrophilic than fatty monoacids, which enhances the solubility of derivatized molecules, reduces their non-specific binding to cell membranes and internalization, thereby better sustaining their in vivo efficacy. Moreover, fatty diacids with sulfonic acid or tetrazole groups at the alkyl terminus also possess high affinity for albumin and can significantly prolong the plasma half-life of derivatized molecules.
In fatty acid derivatization technology, the linker is a key chemical unit that connects the fatty acid to the drug backbone or spacer. Its structure directly affects the physicochemical properties and half-life of the drug. Currently, the most commonly used linker is L-γGlu, whose negative charge helps maintain the affinity between the derivatized molecule and albumin, thereby significantly prolonging the half-life. The tandem use of multiple L-γGlu residues can further enhance the solubility and formulation stability of the drug. Additionally, D-γGlu, butanoyl-sulfonamide, and L-Lys can also serve as effective linkers to achieve half-life extension.
Spacers function by increasing the spatial distance between the fatty acid extension unit and the drug’s active site, thereby reducing steric hindrance caused by albumin binding. This ensures that the drug can still effectively recognize and activate the target receptor even when bound to albumin. Drugs such as semaglutide and tirzepatide commonly employ hydrophilic long-chain spacers (e.g., Ado/AEEA), aiming to maintain sufficient receptor affinity while achieving albumin-mediated long-acting protection.
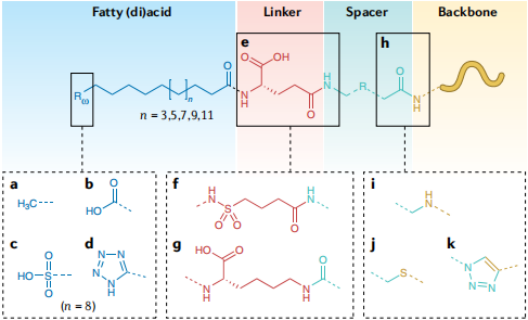
Fig2.Structural Features of Fatty Acid Derivatives
Tide Chem has long been dedicated to the research, development, production, and sales of long-acting side chains for peptide and protein drugs. The company has established a modular compound library comprising hundreds of structurally diverse long-acting side chain molecules. Additionally, Tide Chem offers integrated services such as custom synthesis of long-acting side chains, quality studies, and preparation of CDE submission documents. Leveraging extensive R&D expertise and production capabilities, we have provided a wide range of novel and diversified long-acting side chain products to numerous enterprises worldwide, assisting clients in accelerating the screening and evaluation processes for innovative peptide and protein drug candidates.